Background:Sickle cell illness (SCD) is a bunch of hereditary ailments, inherited as autosomal recessive dysfunction, which causes mutation within the β-globin gene. Because of this, there’s a change within the
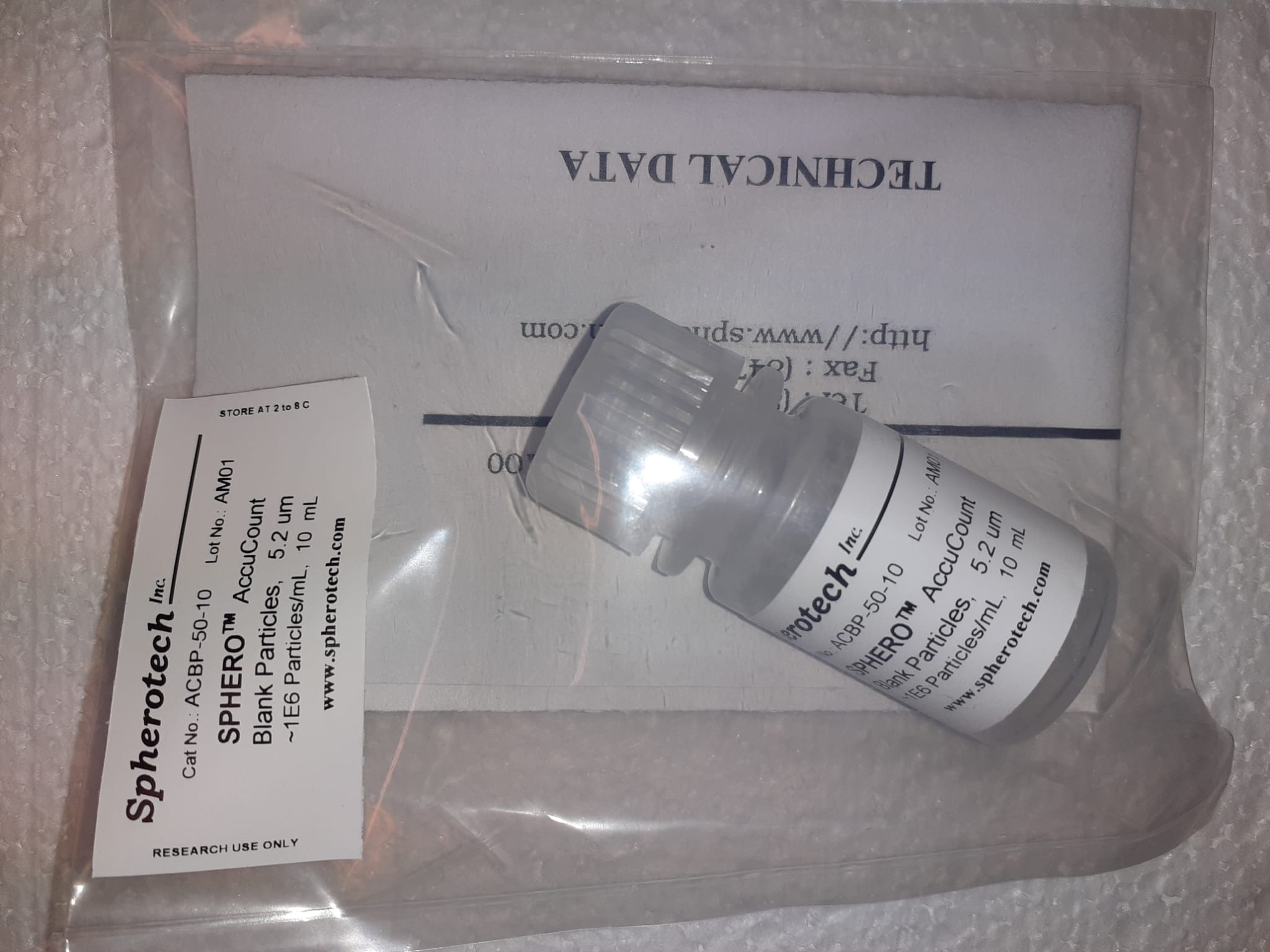
SPHEROTM AccuCount ParticlesSPHEROTM AccuCount Particles
Incorporates particles with a recognized number of particles per mL • Easy to utilize and worth environment friendly • Accessible in quite a lot of sizes to accommodate the aim

Extremely Rainbow Fluorescent ParticlesExtremely Rainbow Fluorescent Particles
Description This product incorporates a single inhabitants of Rainbow Particles that are dyed to a single fluorescent depth. Every Rainbow Particle incorporates a mixture of fluorophores that are stably embedded

MethylFlash Methylated DNA Quantification Equipment (Colorimetric)MethylFlash Methylated DNA Quantification Equipment (Colorimetric)
Product Choices The MethylFlash Methylated DNA Quantification Gear (Colorimetric) is an extra refinement of our earlier Methylamp worldwide DNA methylation quantification kits by simplifying the workflow and enhancing the consistency
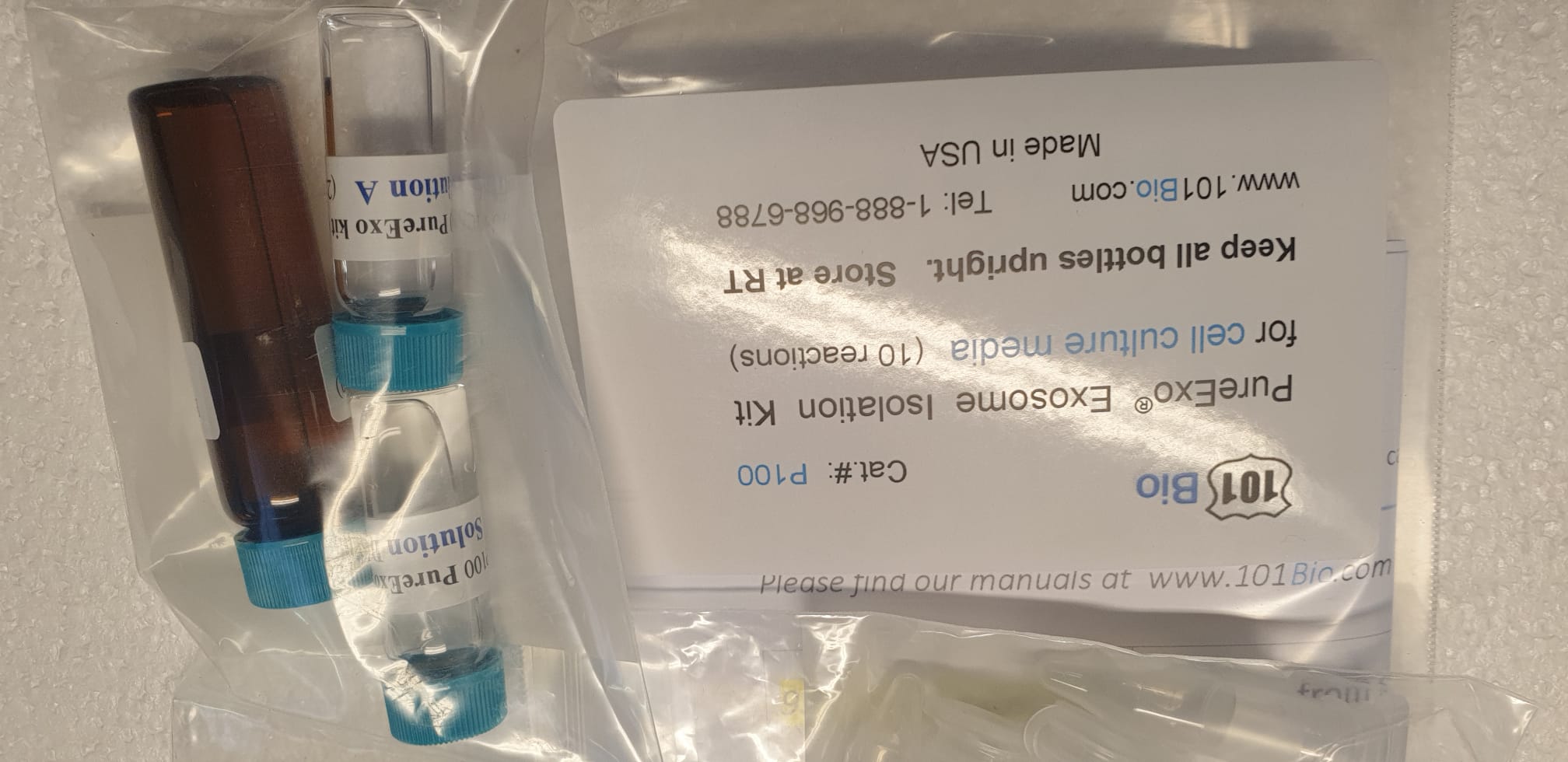
Exosome Isolation Kits purify exosomeExosome Isolation Kits purify exosome
Exosome Isolation Kits purify exosome by affinity purification. Extreme Purity exosomes and completely different Evs (Extracellular vesicles) from the cell custom medium and physique fluid (extreme yield by common microfiltration)

Applied Cell Extracellular MatrixApplied Cell Extracellular Matrix
Utilized Cell Extracellular Matrix In cell biology, processes similar to cell differentiation, cell cycle and cell survival are affected by alerts stimulated by way of cell adhesion. As a result

Oral Administration of Brain Protein Combined With Probiotics Induces Immune Tolerance Through the Tryptophan PathwayOral Administration of Brain Protein Combined With Probiotics Induces Immune Tolerance Through the Tryptophan Pathway
Extreme irritation results in secondary immune harm after traumatic mind damage (TBI). The intestinal mucosa is a key element of immune tolerance because of gut-brain axis regulation, however the healing

Click Chemistry for Imaging in-situ Protein Palmitoylation during the Asexual Stages of Plasmodium falciparumClick Chemistry for Imaging in-situ Protein Palmitoylation during the Asexual Stages of Plasmodium falciparum
Palmitoylation refers back to the modification of the cysteine thiols in proteins by fatty acids, mostly palmitic acid, by means of ‘thioester bond’ formation. In vivo, palmitoylation of proteins is catalyzed

Combining Rapid Microfluidic Mixing and Three-Color Single-Molecule FRET for Probing the Kinetics of Protein Conformational ChangesCombining Rapid Microfluidic Mixing and Three-Color Single-Molecule FRET for Probing the Kinetics of Protein Conformational Changes
Single-molecule Förster resonance vitality switch (FRET) is properly suited to finding out the kinetics of protein conformational modifications, owing to its excessive sensitivity and skill to resolve particular person subpopulations
Rapidly emerging SARS-CoV-2 B.1.1.7 sub-lineage in the United States of America with spike protein D178H and membrane protein V70L mutationsRapidly emerging SARS-CoV-2 B.1.1.7 sub-lineage in the United States of America with spike protein D178H and membrane protein V70L mutations
Summary The SARS-CoV-2 B.1.1.7 lineage is very infectious and as of April 2021 accounted for 92% of COVID-19 instances in Europe and 59% of COVID-19 instances within the U.S.
